Agreement
We use cookies on our website to provide you with the best possible experience. By clicking "Accept All" you agree to the use of all cookies and to our privacy policy.
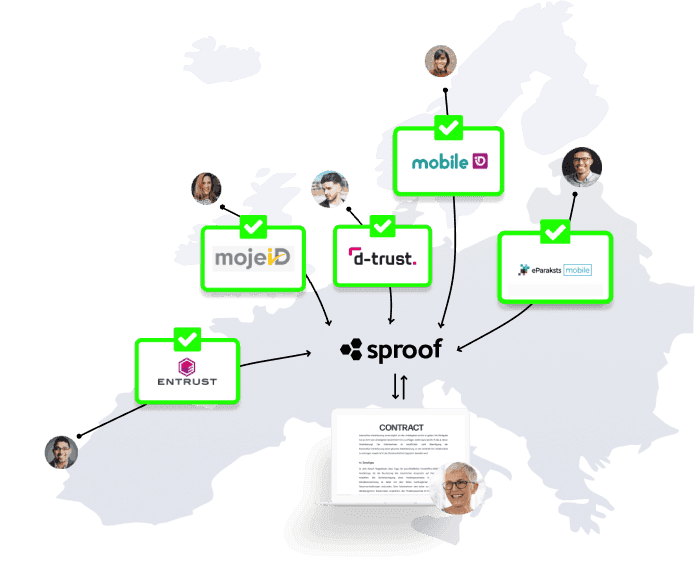
Digital signing requires the highest level of trust and security.
sproof is 100% compliant with national or international regulations and guidelines for digital signatures and digital signature tools.
GDPR
Basic Data Protection Regulation
Data protection is our mission, not just an add-on. sproof is GDPR compliant and meets the requirements of one of the world's most comprehensive legal frameworks for data protection. sproof hosts all of its services within the EU and without the involvement of third countries. Additional customizable automatic deletions and strict technical and organizational measures (TOMs) safeguard the privacy of our customers.FDA
FDA 21 CFR Part 11
sproof sign complies with FDA 21 CFR Part 11. FDA 21 CFR Part 11 is a Code of Federal Regulations that establishes the United States Food and Drug Administration (FDA) regulations on electronic records and electronic signatures (ERES). Part 11 specifically defines the criteria by which electronic records and electronic signatures are considered trustworthy, reliable, and equivalent to paper records.eIDAS
Electronic identification, authentication and trust services
sproof is eIDAS compliant and meets the requirements for legally binding digital signatures according to the EU Regulation on electronic identification and trust services for electronic transactions applicable in Austria, Belgium, Bulgaria, Croatia, Cyprus, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Ireland, Italy, Latvia, Lithuania, Luxembourg, Malta, Netherlands, Poland, Portugal, Romania, Slovakia, Slovenia, Spain and Sweden. sproof sign can also create signatures according to the Swiss Zert-ES for legally binding digital signatures in Switzerland.IÖB
IÖB Award
sproof sign is suitable for public administration and has been awarded by the IÖB. An Austrian expert panel has tested sproof sign and awarded it the IÖB seal, which identifies sproof sign as a tool suitable for the creation and collection of digital signatures for public administration.
"For us, uncompromising data protection is not an add-on, but the foundation of a sovereign solution for the European market"
Dr. Clemens BrunnerCEO & Co-Founder, sproof GmbH

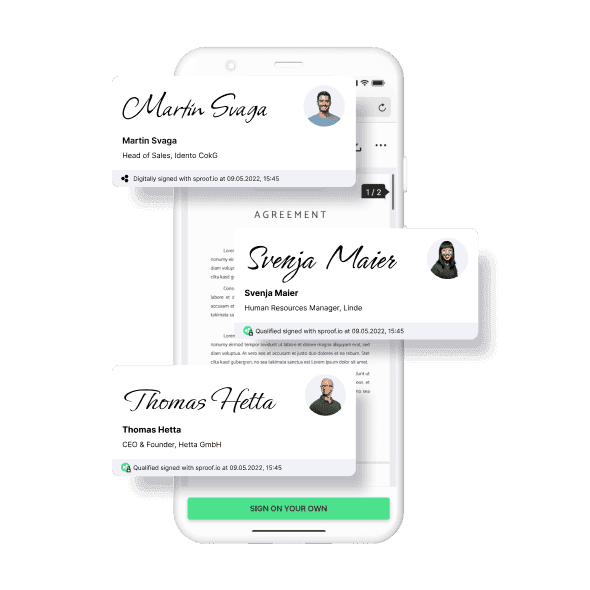
Simple electronic signature (EES)
The simple electronic signature has no probative value and can in no way replace a written signature. In case of agreement of both parties, this method can be a viable option. A common example of a simple signature is an uploaded photo or a scan of a signature placed on a document.
Advanced Electronic Signature (AES)
Every signature made with our platform already achieves the high level of security guaranteed by the advanced electronic signature (AES). The signer is additionally identified, for example, with the help of the e-mail address. Thus, this type of digital signature already has sufficient probative value for many applications.
Qualified Electronic Signature (QES)
According to the eIDAS regulation, the qualified digital signature is legally 100% equivalent to the handwritten signature and meets the highest quality criteria. Therefore, only the qualified signature should be used for contracts with a written form requirement and high demands on the probative value and trust of all contracting parties. sproof has specialized in this type of digital signing.Frequently asked questions
Is the digital signature 100% legally valid?
Are regular penetration tests carried out with sproof sign?
Which e-signature standards does sproof sign offer for your company?
How is IT security ensured? (Are there also documents/certifications for this?)
How long does it take to integrate sproof into existing workflows via API?
Why should companies rely on European tools?
Where are the sproof sign servers located?
Is sproof sign GDPR compliant?
Which legal, security and compliance regulations does sproof sign meet?
Can I also get a qualified signature via sproof sign?
What is the difference between an advanced and a qualified digital signature?
Are signatures created with sproof legally valid?
When do you need a qualified electronic signature (QES)?